empty box
Scroll Up to learn more about this year's supplier-only events.
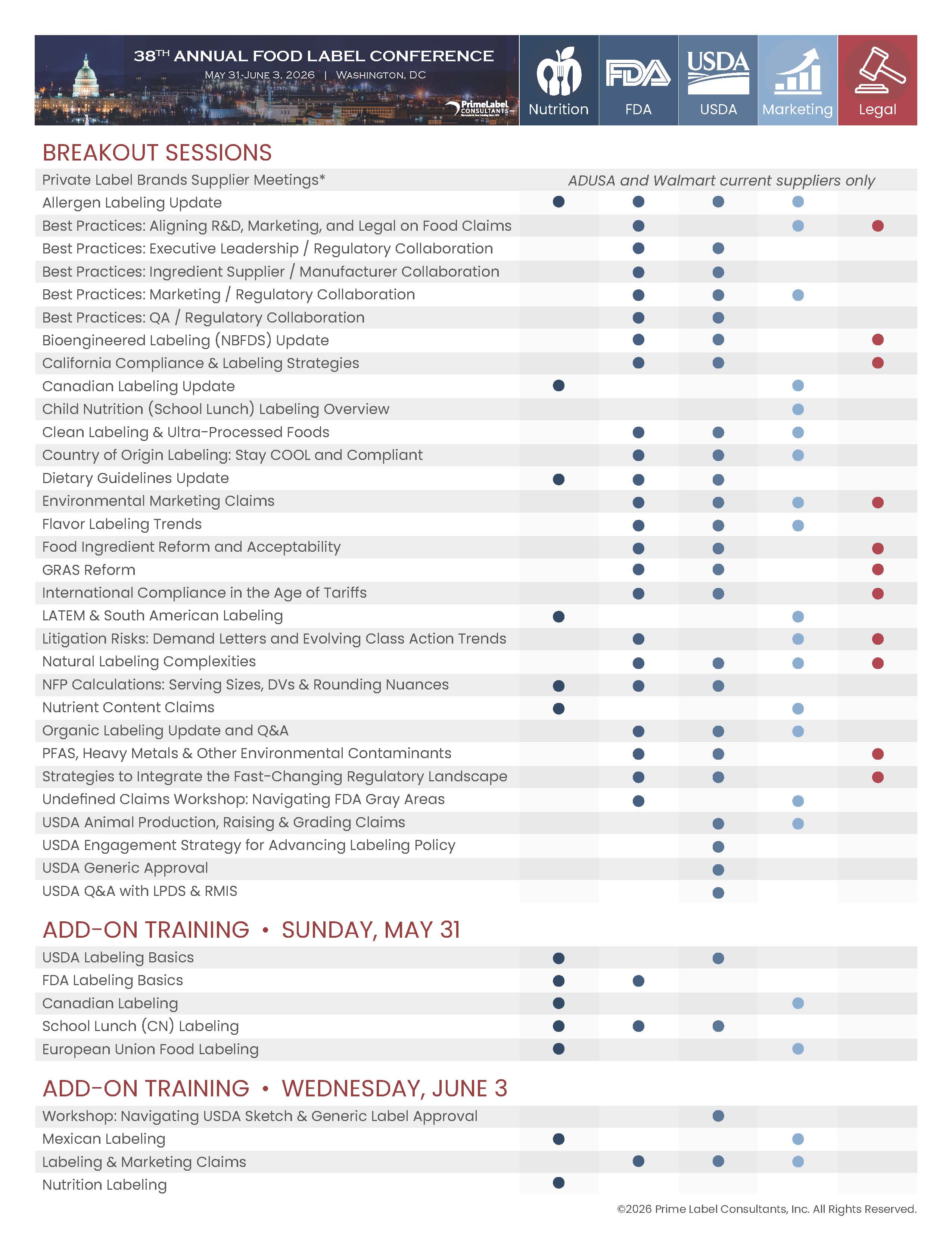
Click Here to view the 2026 Breakout Schedule.
empty box
Allergen Labeling Update
Steven M. Gendel, Gendel Food Safety LLC
Tracks: Nutrition, FDA, USDA, Marketing
Discover the latest updates to Allergen labeling, including all the recent FDA activities this year. Explore the different requirements of the USDA and FDA, and talk through implementation complexities and the dilemmas around when to use precautionary allergen statements.
empty box
Best Practices: Aligning R&D, Marketing, and Legal on Food Claims
Kantha Shelke, Corvus Blue LLC & Ashish Talati, Talati Law
Tracks: FDA, Marketing, Legal
Every food claim starts in R&D and ends on a label, but misalignment across marketing and legal is where innovation stalls and claims fall apart. A food scientist and food attorney team up to share a practical framework for translating ingredient science into substantiated, compliant, and defensible claims—including functional and structure/function positioning—and the cross-functional strategies that keep labels standing up under scrutiny.
empty box
Best Practices: Executive Leadership / Regulatory Collaboration
Sheila Dy Juanco, Upside Foods
Tracks: FDA, USDA
Building on last year's session, this discussion addresses the future of regulatory leadership. The current food labeling landscape—marked by evolving FDA expectations, increased state activity, and heightened risk—means regulatory decisions are now enterprise-level, impacting innovation, brand credibility, and growth. This session will explore how regulatory affairs leaders can be strategic partners, not gatekeepers, guiding teams through uncertainty with risk-based, business-informed decisions. Drawing on real-world experience across emerging food technologies and complex labeling, the discussion will offer practical ways to elevate regulatory influence while maintaining compliance.
empty box
Best Practices: Ingredient Supplier / Manufacturer Collaboration
Jesse Waller, Cargill Protein
Tracks: FDA, USDA
Growth in rapid development and marketing of new product claims has added strain to the ingredient sourcing and regulatory workflow. Building effective and efficient communication paths is necessary to managing this critical coordination. This session will focus on the best practices for relationship management between manufacturer and ingredient supplier and highlight data-driven dependencies and record keeping strategies.
empty box
Best Practices: Marketing / Regulatory Collaboration
Michelle Grow, Melissa's
Tracks: FDA, USDA, Marketing
Marketing and regulatory teams share the same goal—bringing compliant, competitive products to market—but they often speak very different languages. This session explores how organizations can bridge that gap to create stronger product labeling, clearer communication, and more efficient development processes. Through real-world case studies and collaborative strategies, presenters will demonstrate how teams can translate regulatory requirements into marketing opportunities, avoid common pitfalls, and build internal workflows that support both innovation and compliance.
empty box
Best Practices: QA / Regulatory Collaboration
Belinda Davis, Tyson Foods
Tracks: FDA, USDA
Effective collaboration between Regulatory and QA professionals is vital for market compliance and consumer safety. This session explores critical touchpoints where these roles intersect, including raw materials, specifications, labeling and claims, and food safety systems.
empty box
Bioengineered Labeling (NBFDS) Update
Ann M. Begley & Mary Beth Schultz, Wiley Rein LLP
Tracks: FDA, USDA, Legal
With the Ninth Circuit striking down parts of the Bioengineered Labeling rule, questions remain about the impact of this decision, industry considerations and potential outcomes. This session will cover these and other updates and nuances to the specifics of the rule, the options for disclosing BE status and USDA enforcement.
empty box
California Compliance & Labeling Strategies
Mason Weeda & Jennifer Gross, OFW Law
Tracks: FDA, USDA, Legal
This session will discuss several recently announced and key regulatory updates in the state of California. While not an all-encompassing list, this presentation will discuss, additive bans, date labeling provisions, state Executive Orders, trends in Proposition 65 litigation of particular interest to the food industry, and baby food testing disclosure requirements. This session will provide timely, valuable insights and practical advice for staying compliant and achieving business goals in the evolving landscape of CA food law.
empty box
Canadian Labeling Update
Michelle Anstey, NSF International
Tracks: Nutrition, Marketing
Understand new Canadian nutrition and ingredient labeling regulations and learn how they differ from labeling in the US, including the Front-of-Package symbol, Nutrition Facts Table and ingredient list formats, and allergen declarations. Discuss the subtleties of formulating products for Canada and some of the special issues that apply. Understand what resources are available to you to ensure your Canadian labels are compliant.
empty box
Child Nutrition (School Lunch) Labeling Overview
Patricia Phillips, Phillips Resources
Tracks: Marketing
Join a leading expert in Child Nutrition labeling for an overview of program crediting and the application process. Explore how CN labeling works and discuss current trends and what's on the horizon. Learn about the Agricultural Marketing Service (AMS) quality control program and the CN label application process. Find out where to obtain information and discuss some frequently asked questions.
empty box
Clean Labeling & Ultra-Processed Foods
Kantha Shelke, Corvus Blue LLC
Tracks: FDA, USDA, Marketing
Ultra-processed (UPFs) foods face growing consumer scrutiny, often driven by fear-based narratives and misinformation. This session will separate the perception of UPFs and clean labeling from scientific reality, debunk common myths, and explore the industry’s role in informed decision-making. It will also examine the implications for food labeling and potential litigation, equipping attendees with a science-based perspective to navigate this evolving landscape.
empty box
Country of Origin Labeling: Stay COOL and Compliant
Sonja Jones, Food Disclosure and Labeling Division, USDA AMS
Tracks: FDA, USDA, Marketing
Country of Origin Labeling is managed by the Food Disclosure and Labeling Division of AMS USDA, and required for retail labeling of a range of raw agricultural commodities. Discover the essentials of USDA’s COOL requirements, including covered commodities, labeling formats, and compliance strategies. Through real-world examples and an interactive FAQ, you’ll gain practical insights to keep your labels accurate and your business compliant.
empty box
Dietary Guidelines Update
Grace Chamberlin, Center for Science in the Public Interest (CSPI)
Tracks: Nutrition, FDA, USDA
The 2025 Dietary Guidelines are moving forward under new leadership at HHS, with Secretary Robert F. Kennedy Jr. pushing a shift toward shorter, more accessible guidance and an emphasis on whole foods. His comments and the new DGA's guidance on dairy, saturated fat, protein, and "highly processed" foods have sparked both interest and questions about how science, policy, and industry align in the final publication. This session will explore the original advisory committee’s report, the new "Scientific Foundation" used in this update process, and what changes could mean for food companies, health professionals, and consumers alike.
empty box
Environmental Marketing Claims
Katie Bond, Keller and Heckman LLP
Tracks: FDA, USDA, Marketing, Legal
This session will provide an overview of the current legal landscape for green marketing by focusing on five of the most significant developments affecting food companies right now – from class actions, to state enforcement, National Advertising Division cases, and the roll-out of the FTC’s revised Green Guides.
empty box
Flavor Labeling Trends
Lisa Cummins, McCormick Flavor Solutions | McCormick FONA
Tracks: FDA, USDA, Marketing
Flavor labeling tops the charts as one of the most common complicated aspects of product labeling. This session will cover flavor descriptors and the laws governing labeling and specific situations including food allergens, flavoring agents, ingredient classifications, flavor categories, organic, kosher and halal.
empty box
Food Ingredient Reform and Acceptability
Mason Weeda & Eric J. Steiner, OFW Law
Tracks: FDA, USDA, Legal
This session will include an overview of food ingredient reforms that are occurring at the state and federal level. These include ingredient bans, labeling – including warnings and front of pack, generally recognized as safe requirements, school lunch programs, and SNAP waivers. With the state patchwork in mind, this session will also discuss litigation in certain states and efforts at the federal level towards national uniformity.
empty box
GRAS Reform
Evangelia Pelonis & Tony Pavel, Keller and Heckman LLP
Tracks: FDA, USDA, Legal
The FDA’s “Generally Recognized as Safe” (GRAS) program is under review, with HHS directing the agency to consider eliminating the self-affirmed GRAS pathway. At the same time, Congress is considering the Ensuring Safe and Toxic-Free Foods Act of 2025, which would require mandatory GRAS notifications, public input, and regular chemical reassessments. The FDA has also begun a proactive post-market review framework while continuing to issue “no questions” letters for new substances. This session will provide an overview of these policy developments and their potential implications for industry.
empty box
International Compliance in the Age of Tariffs
Katelyn M. Hilferty & Casey Weaver, Morgan, Lewis & Bockius LLP
Tracks: FDA, USDA, Marketing, Legal
US trade policy under the new administration is well underway, with headlines about US tariffs, global retaliatory tariffs, and US retaliatory tariffs crossing the news on a near daily basis. Many companies who historically have not had to manage tariff risks—because they operate in nations with which the US has a trade agreement, do not take on direct US import risks, or operate in a primarily domestic market—are finding duties and tariffs among the top of their internal risk factors. In this session led by Morgan, Lewis & Bockius International Trade partners Katelyn (Katie) Hilferty and Casey Weaver, we will discuss recent tariff developments, compliance strategies, and practical tips for mitigating tariff impact.
empty box
LATEM & South American Labeling
Phil Daniel, TechLink International
Tracks: Nutrition, Marketing
A critical session if you are exporting food to Latin or South America. TechLink will share thier overview of the current food labeling standards, with a focus on general requirements, including front of package requirements, stop signs, nutritional panel calculations and more.
empty box
Litigation Risks: Demand Letters and Evolving Class Action Trends
Marshall L. Baker, Akin Gump
Tracks: FDA, Marketing, Legal
Plaintiff litigation and demand letters are becoming one of the primary risks to consider in developing product marketing strategies. Learn how to navigate these risks with a discussion of the current litigation landscape, how to best protect your company, and what to do if your product becomes a target.
empty box
Natural Labeling Complexities
Evangelia Pelonis, Keller and Heckman LLP
Tracks: FDA, USDA, Marketing, Legal
The natural products market continues to grow in the US and around the globe. This session is critical to understand the ins and outs of Natural claims on labeling. Explore the differences between FDA and USDA definitions of “Natural”, and when the claim may present litigation risks. Discuss the impact on retail, restaurants and customer perception, and explore the pros and cons of this contentious claim, both on the package and in related advertising and marketing.
empty box
NFP Calculations: Serving Sizes, DVs & Rounding Nuances
Fred Mosher & Michelle Liang, Prime Label Consultants
Tracks: Nutrition, FDA, USDA
Explore the integrity of the Nutrition Panel by understanding the differences between calculated and analytical methods of producing nutrient data. Discuss the various sources of nutrient data and how to apply them to determine serving size and daily values. Understand the nuances of FDA rounding rules. Includes a brief introduction to Prime Label’s EZ FormⓇ food labeling software.
empty box
Nutrient Content Claims
Fred Mosher, Prime Label Consultants
Tracks: Nutrition, Marketing
Learn about FDA and USDA requirements for expressed and implied claims including common call-outs such as Low Calorie, Low Fat, No Added Sugar and High Protein. Explore relative claims and front of pack nutrient claims and the subtleties of their different requirements.
empty box
Organic Labeling Update and Q&A
Erin Healy, National Organic Program, USDA AMS
Tracks: FDA, USDA, Marketing
This session, led by the National Organic Program, will cover the latest regulatory updates to the organic standards and explore key industry trends. This presentation offers essential insights for retailers, followed by an interactive Q&A.
empty box
PFAS, Heavy Metals & Other Environmental Contaminants
Brian P. Sylvester, Morrison Foerster
Tracks: FDA, USDA, Legal
Contaminants may emanate from any direction. This presentation will focus on prevention, detection strategies, and best practices to avoid heavy metal contaminants from coming through unknowingly in imported ingredients. We will also explore how packaging components and public water systems can add another complication to the mix, highlighting the breadth of PFAS contamination and its impact on litigation in the years ahead. It is difficult to test for every conceivable contaminant. Those attending this session will come away with additional strategies for reducing liability and risks to the food supply.
empty box
Strategies to Integrate the Fast-Changing Regulatory Landscape
Amaru J. Sanchez, Akin Gump
Tracks: FDA, USDA, Legal
Building an effective compliance program increasingly requires companies to navigate and align fast‑moving regulatory and legal developments at both the federal and state levels. As nutrition, ingredient, and labeling requirements continue to expand and evolve, organizations face growing pressure to understand what is changing—and how those changes fit together. This session will explore practical strategies for synthesizing these developments into a cohesive, forward‑looking compliance framework that helps companies manage risk while remaining agile in a shifting regulatory environment.
empty box
Undefined Claims Workshop: Navigating FDA Gray Areas
Caitlin Diederich, Prime Label Consultants
Tracks: FDA, Marketing
This workshop offers a framework for assessing undefined claims. Discuss how to consider regulatory definitions, pending actions, industry standards and enforcement risks to make informed decisions. Explore definitions in the form of brand standards vs. third-party certification. Simulate consumer responses to common claims to practice identifying truthful vs. misleading messaging. Walk away with an appreciation - the bigger the claim, the bigger the burden of proof!
empty box
USDA Animal Production, Raising & Grading Claims
Emily Hendricks, Prime Label Consultants
Tracks: USDA, Marketing
Discuss the complexity of Animal Production, Raising & Breed claims such as Cage-Free Poultry, Free-Range, No Added Hormones or Antibiotics, Humanely Raised, Certified Angus and others. Find out the latest in USDA grading claims.
empty box
USDA Engagement Strategy for Advancing Labeling Policy
Jeffery Canavan, OFW Law
Tracks: USDA
Learn how to navigate the complex regulatory landscape by engaging directly with FSIS Office of Policy and Program Development (OPPD) experts on innovative labeling and emerging food technologies. This session provides practical guidance on submitting inquiries through formal channels to ensure your novel products meet federal regulatory and compliance standards.
empty box
USDA Generic Approval
Emily Hendricks, Prime Label Consultants
Tracks: USDA
Find out the current regulations, policies, and best practices for generic approval. Explore generic eligibility, including changes that can be made to previously approved labels. Understand generic approval compliance, recordkeeping requirements, and discuss strategies to reduce risk of enforcement.
empty box
USDA Q&A with LPDS
Team members of the Labeling and Program Delivery Staff, FSIS-USDA
Team members of the Risk Management and Innovations Staff, FSIS
Tracks: USDA
Explore the ins and outs of USDA labeling by bringing your specific questions on the subtleties of labeling to USDA's Labeling and Program Delivery Staff (LPDS).